Education:
BS, The State University of New York at Oswego, 1995
PhD, University at Buffalo, The State University of New York, 2001
Professional Experience:
Associate Professor, University of Missouri-Columbia, 2017-present
Assistant Professor, University of Missouri-Columbia, 2011-2017
Research Scientist, Oak Ridge National Laboratory, 2005-2010
Postdoctoral Fellow, Los Alamos National Laboratory, 2001-2005
Selected Professional Activities:
Editor in Chief, Inorganic and Nano-Metal Chemistry, 2025-present
Associate Editor, Green Chemistry Letters and Reviews, 2021-present
Honors and Awards:
H-index: 83
MU Graduate Faculty Mentor Award, 2024
Distinguished Alumnus Award, State University of New York, Oswego, 2023
Gold Chalk Award, Graduate Professional Council, 2020
Fuldner Faculty Fellow, MU Department of Chemistry, 2019 & 2020
Cottrell Scholar Award, 2015
Provost's Outstanding Junior Faculty Research and Creative Activity Award, 2015
George W. Thorn Award, University at Buffalo Distinguished Alumni Award, 2010
Presidential Early Career Award for Scientists and Engineers (PECASE), U.S. Department of Energy, 2008
Office of Science Outstanding Mentor Award, U.S. Department of Energy, 2007
Eugene P. Wigner Fellowship, Oak Ridge National Laboratory, 2005-2007
Frederick Reines Postdoctoral Fellowship, Los Alamos National Laboratory, 2002-2005
American Chemical Society Division of Analytical Chemistry Graduate Fellowship, 2000
Society for Applied Spectroscopy Graduate Student Award, 1999

Baker Group Spring 2026
Light-driven nanochemistry; Nanocatalysis; Nanoparticle-based theranostics and bioimaging; Water cleanup; Waste valorization; (Bio)analytical and environmental sensors; Deep eutectic solvents & ionic liquids; nano- and microplastics; photothermal and photoacoustic agents
Our work is motivated and inspired by the United Nations' Sustainable Development Goals (17):
- No poverty
- Zero hunger
- Good health and well-being
- Quality education
- Gender equality
- Clean water and sanitation
- Affordable and clean energy
- Decent work and economic growth
- Industry, innovation, and infrastructure
- Reduced inequalities
- Sustainable cities and communities
- Responsible consumption and production
- Climate action
- Life below water
- Life on land
- Peace, justice, and strong institutions
- Partnerships for the goals
Research in the Baker Group is highly cross-disciplinary and collaborative in nature and is characterized by problem-solving using sustainable nanoscience and task-specific solvent-engineering approaches. Examples of ongoing research projects include:
- Synthetic nanoclays with programmable surface chemistry
- Plasmon-driven synthesis and photocatalysis
- Eutectogels: soft materials for drug release, wastewater treatment, and on-skin electronics
- Fluorescent, theranostic, catalytic, and photoacoustic nanomaterials
- Advanced materials for water purification (PFAS, micro- and nanoplastics) & desalination
- Bioimaging agents (e.g., myelin imaging, cancer detection)
- Waste valorization (e.g., upcyling food and plastic waste to useful materials)
- Benign-by-design solvents (ionic liquids, deep eutectic solvents, zwitterionic liquids)
- Deep eutectic media for performing materials synthesis (e.g., perovskite quantum dots)
- Promoting diversity, equity, inclusion and opportunity in the classroom and the laboratory
Women in Training for Science (WITS)
In partnership with Dr. Kathryn Fishman-Weaver—formerly, an award-winning secondary gifted education teacher at Rock Bridge High School—we launched an outreach program in 2013 with the overarching goal of cultivating the talents of female students from the high school to graduate school level, with seminars, hands-on learning activities, laboratory experiences, supervised training, sibling proxy (“big sister”) mentorship, and internships designed to ignite and promote a lifelong interest in science and scientific careers. For general inquiries, or for those interesting in participating, please feel free to contact me at: bakergar@missouri.edu.
(selected from a list of over 340 articles; for a complete publication listing, please refer to my Google Scholar page)
Ogbodo, R., Adhikari, L.; Adu, C.; Nieuwkoop, A. J.; Baker, G. A.; Wishart, J. F.; Margulis, C. J. Charge Tethering Drives Intermediate-Range Order and Slow Dynamics in Zwitterionic Liquids. 2026, submitted.
Stettler, A. M.; Blanchard, S. S.; Baker, G. A.; Blanchard, G. J. The Direct Piezoelectric Effect in Deep Eutectic Solvents. J. Am. Chem. Soc. 2026, 148, 5900–5904. (link)
Bhawawet, N.; Chen, X.; Ballou-Crawford, J. J.; Polo-Parada, L.; Baker, G. A. Smart Ionic Liquid Sensory Ink for Ratiometric Fluorescence Temperature Surface Mapping. J. Ionic Liq. 2026, 6, 100203. (link)
Akter, S.; Kamoldinov, A.; Roy, A.; Baker, G. A.; Haider, M. R. A Vision Driven Framework for Droplet Monitoring and Feature Extraction in Inkjet Printing. IEEE Sens. Lett. 2026, submitted.
Li, Q.; Hasan, Md. Z.; Dipti, T.; Qin, H.; Skidmore, J.; Guo, A.; Baker, G. A.; Liu, L.; Feng, L.; Yang, Z. Material-Induced Selectivity Shift: Tuning Cleavage Preference of Serine Proteases via Confinement in Metal-Organic Materials. ACS Appl. Bio Mater. 2026, submitted.
Laber, C. H.; Zheng, S. Kimble, A.; Bednar, A.; Fang, J.; Wei, T.; Baker, G. A. Hydrophobic Deep Eutectic Solvents for the Efficient Removal of Priority Polyfluoroalkyl Substances from Water. 2026, submitted.
Saha, M.; Saraswat, J.; Baker, G. A.; Behera, K.; Trivedi, S. Tunable Disruption of Pseudoisocyanine J-Aggregates by Choline Chloride-Based Deep Eutectic Solvents. Phys. Chem. Chem. Phys. 2026, accepted.
Tupkar, H.; McAlpine, J.; Puls, T. D. P.; Ishtaweera, P.; Baker, G. A.; Gebbie, M. A. Utilizing Oxygenated Functionality to Localize Charge and Increase Capacitance in Aromatic Ionic Liquids. ACS Electrochem. 2026, in revision.
Ishtaweera, P.; Larm, N. E.; Baker, G. A. Pyrene-Tagged Polyimidazolium Nanoclays Enable Modular Excimer Emission for Multi-Analyte Fluorescence Sensing. ACS Appl. Nano Mater. 2026, in press.
Samaranayake, A.; Roy, A.; Polo-Parada, L.; Laber, C. H.; Baker, G. A. Light- and Dark-Directed Morphological Control in N-Methyl-D-Glucamine-Mediated Gold Nanostar Synthesis: Implications for Photothermal Performance. ACS Appl. Nano Mater. 2026, in press.
Jha, M. K.; Ishtaweera, P.; Dietert, C.; Bañuelos, J. L.; Baker, G. A.; Kashyap, H. K. Temperature Dependence of Structural Landscape of Thymol-Coumarin Based Hydrophobic Deep Eutectic Solvent as Revealed by X-ray Scattering and Molecular Simulations. J. Phys. Chem. B 2026, in revision.
Samaranayake, A.; Baker, G. A. Phospho-L-ascorbic acid: A Mild and Versatile Reducing and Capping Agent for Gold Nanoparticle Synthesis. Green Chem. Lett. Rev. 2026, in revision.
Roy, A.; Wallinger, J.; Ishtaweera, P.; Dzorkpata, C.; Larm, N. E.; Zhu, P.; Baker, G. A. Enhanced Photoluminescence and Optoelectronic Properties of Blue-Emitting CsPbCl3 Nanocrystals via Deep Eutectic Solvent Synthesis. 2026, submitted.
MacDonald, R. T.; Siegel, A. L.; Pokpas, K.; Baker, G. A.; Iwuohaa, E.; Cupido. C. Photo-induced Silver Shell Growth on Gold Nanostars for Enhanced Electrochemical Functionality. Appl. Surf Sci. 2026, 720, 165381. (link)
Fakayode, S. O.; Baker, G. A.; Bwambok, D. K.; Brady, P. N.; Ishtaweera, P.; Benjamin, U. Host−Guest Cyclodextrins and Cyclodextrin-Fluorescence Sensors: Applications in Environmental, Pharmaceutical, Biomedical Research and Clinical Diagnosis: A Review. Appl. Spectrosc. Rev. 2025, 1–66. (link)
Dipti, T. T.; Hasan, M. Z.; Armstrong, Z.; Quadir, M.; Liu, L.; Wan, C.; Baker, G. A.; Feng, L.; MacRae, A.; Yang, Z. Water as An Eco-Friendly Liquid-Assisted Grinding Agent (ecoLAGent) to Expand Metal-Organic Framework “Library” for Green Enzyme Encapsulation. ACS Appl. Mater. Interfaces 2025, 17, 55615–55627. (link)
Zhao, H.; Roy, A.; Samaranayake, A.; Ishtaweera, P.; Baker, G. A.; Duong, N.; Markmann, L. G.; Fernando, N. S.; Mitchell-Koch, K. R. Lipase-Catalyzed Michael Addition in ‘Water-like’ Ionic Liquids and Tertiary Amides: What is the Role of Enzymes? Langmuir 2025, 41, 12718–12730. (link)
Stettler, A.; Baker, G. A.; Blanchard, G. J. The Importance of Hydrogen Bonded Networks in the Dynamic Heterogeneity of Deep Eutectic Solvents. J. Phys. Chem. B 2025, 129, 6300–6308. (link)
Olusegun, S. J.; Blanchard, S. S.; Baker, G. A.; Blanchard, G. J. Quantitation of the Piezoelectric Coefficients in Room Temperature Ionic Liquids. J. Chem. Phys. 2025, 163, 054505. (link)
Stettler, A.; Ishtaweera, P.; Baker, G. A.; Blanchard, G. J. Prominent Composition-Dependent Dynamics Decoupling in the Choline Chloride-Glycerol Deep Eutectic Solvent System. Appl. Spectrosc. 2025, 79, 1325-1333. (link)
Turner, Z.; Laber, C. H.; Kelley, S. P.; Jenness, G.; Roy, A.; Polo-Parada, L.; Baker, G. A. Tuning Photothermal Properties through Donor-Acceptor Engineering: PD:TTF versus TCNQ:TTF Cocrystals. ACS Appl. Energy Mater. 2025, in press. (link)
Álvarez-Martínez, J. U. U.; Castro-Beltrán, R.; Gutiérrez-Juárez, G.; Baker, G. A.; Polo-Parada, L. Microscale Photoacoustic Imaging via Laser-Induced Ultrasound in Microfluidic Environments. J. Phys. Photonics 2025, in press. (link)
Ishtaweera, P.; Larm, N. E.; Polo-Parada, L.; Baker, G. A. Programmable Fluorescent Polyionic Nanoclays as Sensory Materials. Chem. Mater. 2025, 37, 857–868. (link)
Larm, N. E.; Stachurski, C. D.; Trulove, P. C.; Tang, X.; Shen, Y.; Durkin, D. P.; Baker, G. A. The Role of Heavy Water in the Synthesis and Nanocatalytic Activity of Gold Nanoparticles. ACS Nanoscience Au 2025, 5, 52–59. (link)
Sikligar, K.; Patil, R. S.; Zhang, C.; Kelley, S. P.; Ishtaweera, P.; Wagle, D. V.; Barnes, C. L.; Baker, G. A.; Atwood, J. L. Nanotoroids or Coordination Networks: Molecular Constructs at the Mercy of Anions. Inorg. Chem. 2025, 64, 4884–4891. (link)
Musozoda, M.; Bishuk, A. L.; Britton, B. J.; Scheuren, M.; Laber, C. H.; Baker, G. A.; Baker, M.; Zeller, M.; Paull, D. H.; Hillesheim, P. C.; Mirjafari, A. Property-Driven Design of Thermally Robust Organophosphorus Ionic Liquids for High-Temperature Applications. ACS Appl. Eng. Mater. 2025, 3, 1468–1482. (link)
Kaur, G.; Hedouin, G.; Mandal, R.; Jasinski, J. B.; He, X.; Su, M.; Liu, J.; Guo, X.; Walensky, J.; Baker, G. A.; Gallou, G.; Handa, S. Micelle-Dependent Spontaneous Formation of Gold(I) in Nanodendritic Chloride-Bridged Particles with Catalytic Activity for Cyclization of Alkynylanilines in Aqueous Environment. ACS Catalysis 2024, 14, 15645–15653. (link)
Stettler, A.; Ishtaweera, P.; Baker, G. A.; Blanchard, G. J. Molecular-Scale Interactions in the Choline Chloride-Ethylene Glycol Deep Eutectic Solvent System. The Importance of Chromophore Charge in Mediating Rotational Dynamics. J. Phys. Chem. B 2024, 128, 9536–9543. (link)
Ishtaweera, P.; Ray, C. L.; Filley, W.; Cobb, G.; Baker, G. A. Nanoplastics Extraction from Water by Hydrophobic Deep Eutectic Solvents. ACS Appl. Eng. Mater. 2024, 2, 1460–1466. (link)
Dasanayake, G. S.; Hamadani, C. M.; Singh, G.; Kumar Misra, S.; Sharp, J. S.; Adhikari, L.; Baker, G. A.; Tanner, E. E. L. Imidazolium-based Zwitterionic Liquid-modified PEG-PLGA Nanoparticles as Intravenous Drug Delivery Carriers. Nanoscale 2024, 16, 5584–5600. (link)
Fakayode, S. O.; Mehari, T. F.; Fernand Narcisse, V. E.; Grant, C.; Taylor, M. E.; Baker, G. A.; Siraj, N.; Bashiru, M.; Denmark, I.; Oyebade, A.; Bwambok, D. K.; Kuedukey, C.; Alonge, T.; Anum, D. Microplastics: Challenges, Toxicity, Spectroscopic and Real-time Detection Methods. Appl. Spectrosc. Rev. 2024, in press. (link)
Gremillion, A.; Ross, J.; Yu, X.; Ishtaweera, P.; Anwander, R.; Autschbach, J.; Baker, G. A.; Kelley, S. P.; Walensky, J. R. Facile Oxidation of Ce(III) to Ce(IV) Using Cu(I) Salts. Inorg. Chem. 2024, 63, 9602–9609. (link)
Roy, A.; Healey, C. P.; Larm, N. E.; Ishtaweera, P.; Roca, M.; Baker, G. A. The Huge Role of Tiny Impurities in Nanoscale Synthesis ACS Nanoscience Au 2024, 4, 176–193. (link)
Siegel, A. L.; Mallott, A. J.; Patel, D. N.; Polo-Parada, L.; Baker, G. A. Silver(I)-Induced Anisotropy in Plasmon-Driven Growth of Nanoscale Gold. Green Chem. Lett. Rev. 2024, 17, 2306816. (link)
Hossain, M. I.; Wang, H.; Adhikari, L.; Baker, G. A.; Mezzetta, A.; Guazzelli, L.; Mussini, P.; Xie, W.; Blanchard, G. J. Structure-Dependence and Mechanistic Insights into the Piezoelectric Effect in Ionic Liquids. J. Phys. Chem. C 2024, 128, 1495–1505. (link)
Boogaart, D. J.; Baker, G. A. Moving Beyond Choline: Protic Choline Iodide Analogs Toward Cosolvent-Free, Self-Contained Deep Eutectic Electrolytes for Dye-Sensitized Solar Cells. ACS Appl. Eng. Mater. 2024, 2, 360–367. (link)
Ishtaweera, P.; Baker, G. A. Progress in the Application of Ionic Liquids and Deep Eutectic Solvents for the Separation and Quantification of Per- and Polyfluoroalkyl Substances. J. Hazard. Mater. 2024, 465, 132959. (link)
Fakayode, S. O.; Lisse, C.; Medawala, W.; Brady, P. N.; Bwambok, D.; Anum, D.; Alonge, T.; Taylor, M. E.; Baker, G. A.; Mehari, T.; Rodriguez, J. D.; Elzey, B.; Siraj, N.; Macchi, S.; Le, T.; Forson, M.; Bashiru, M.; Narcisse, V. E. F.; Grant, C. Fluorescent Chemical Sensors: Applications in Analytical, Environmental, Forensic, Pharmaceutical, Biological, and Biomedical Sample Measurement and Clinical Diagnosis. Appl. Spectrosc. Rev. 2024, 59, 1–89. (link)
Borah, B.; Acharya, G. R.; Grajeda, D.; Emerson, M. S.; Harris, M. A.; Abeykoon, A. M. M.; Sangoro, J.; Baker, G. A.; Nieuwkoop, A. J.; Margulis, C. J. Do Ionic Liquids Slow Down in Stages? J. Am. Chem. Soc. 2023, 145, 25518–25522. (link)
Sikligar, K.; Kelley, S. P.; Baker, G. A.; Atwood, J. L. Hierarchical 2D Honeycomb-like Network from Barium-Seamed Nanocapsules. Chem. Commun. 2023, 59, 14591-14594. (link)
Bhawawet, N.; Polo-Parada, L.; Ishtaweera, P.; Larm, N. E.; Baker, G. A. Purposefully Designed Surfactants for Facile and Controllable Gold Colloidal Nanocrystal Synthesis. ACS Omega 2023, 8, 41633–41640. (link)
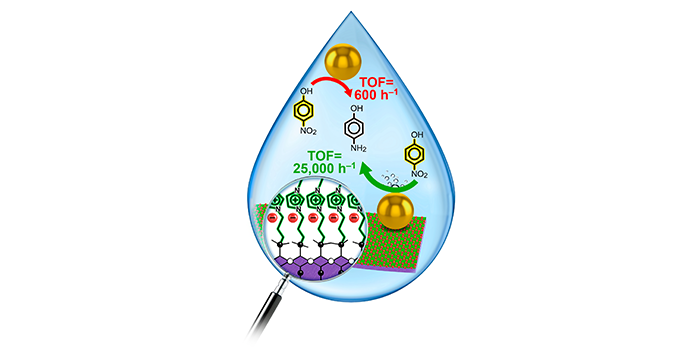
Larm, N. E.; Wagle, D. V.; Ishtaweera, P.; Roy, A.; Baker, G. A. Surface Programmable Polycationic Nanoclay Supports Yielding 100,000 per Hour Turnover Frequencies for a Nanocatalyzed Canonical Nitroarene Reduction. ACS Appl. Eng. Mater. 2023, 1, 1913–1923. (link)
Sikligar, K.; Kelley, S. P.; Wagle, D. V.; Ishtaweera, P.; Baker, G. A.; Atwood, J. L. Nanocapsules of Unprecedented Internal Volume Seamed by Calcium Ions. Chem. Sci. 2023, 14, 9063-9067. (link)
Larm, N. E.; Roy, A.; Ishtaweera, P.; Adhikari, L.; Baker, G. A. Selective Aerobic Oxidation of Benzyl Alcohol under Ambient Conditions using Polyionic Nanoclay-Supported Gold/Palladium Nanoparticles. Mol. Catal. 2023, 546, 113237. (link)
Siegel, A. L.; Adhikari, L.; Salik, S.; Baker, G. A. Progress and Prospects for Deep Eutectic Solvents in Colloidal Nanoparticle Synthesis. Curr. Opin. Green Sustain. Chem. 2023, 41, 100770. (link)
Daykin, A. A.; Ravula, S.; Kaiser, H.; Heitmann, T.; Sanjeewa, L. D.; Baker, G. A.; He, X.; Mazza, A. R.; Miceli, P. F. Disorder and Hydrogenation in Graphene Nanopowder Revealed by Complementary X-ray and Neutron Scattering. Carbon 2023, 206, 277–285. (link)
Hossain, M. I.; Adhikari, L.; Baker, G. A.; Blanchard, G. J. Relating the Induced Free Charge Density Gradient in a Room Temperature Ionic Liquid to Molecular-Scale Organization. J. Phys. Chem. B 2023, 127, 1780–1788. (link)
Anderson, G. I.; Hardy, D.; Hillesheim, P. C.; Wagle, D. V.; Zeller, M.; Baker, G. A.; Mirjafari, A. Anticancer Agents as Design Archetypes: Insights into the Structure–Property Relationships of Ionic Liquids with a Triarylmethyl Moiety. ACS Phys. Chem Au 2023, 3, 94–106. (link)
Zhao, H.; Baker, G. A. Functionalized Ionic Liquids for CO2 Capture under Ambient Pressure. Green Chem. Lett. Rev. 2023, 16, 2149280. (link)
Siegel, A. L.; Polo-Parada, L.; Baker, G. A. Plasmon-Controlled Shaping of Gold Nanostar Photothermal Therapy Agents. Chem. Commun. 2022, 58, 13119–13122. (link)
Steinert, R. M.; Heikes, M. E.; Mitchell-Koch, J. T.; Baker, G. A.; Mitchell-Koch, K. R. Complexometric Titration of Bismuth in Over-the-Counter Stomach Relief Products. J. Chem. Educ. 2022, 99, 3723–3730. (link)
Wang, Y.; Adhikari, L.; Baker, G. A.; Blanchard, G. J. Cation Structure-Dependence of the Pockels Effect in Aprotic Ionic Liquids. Phys. Chem. Chem. Phys. 2022, 24, 18067–18072. (link)
Wang, Y.; Adhikari, L.; Baker, G. A.; Blanchard, G. J. Characterizing the Structure-Dependence of the Induced Free Charge Density Gradient in Imidazolium and Pyrrolidinium Ionic Liquids. Phys. Chem. Chem. Phys. 2022, 24, 19314–19320 (link).
Boogaart, D. J.; Essner, J. B.; Baker, G. A. Halide Effects on the Performance of Equimolar Choline Halide : Guanidinium Thiocyanate Deep Eutectic Solvents as Dye-Sensitized Solar Cell Electrolytes. Green Chem. Lett. Rev. 2022, 15, 615–626. (link)
Kist, J. A.; Essner, J. B.; Woodward, J. D.; Baker, G. A. Silver-Mediated Squaric Acid Reduction as a Facile, Ambient-temperature and Seedless Route to Tunable Bimetallic Au/Ag Nanostars and Nanosnowflakes. ChemNanoMat 2022, 8, e202200189. (link)
Essner, J. B.; Boogaart, D. J.; Baker, S. N.; Baker, G. A. Effects of Carbon Nanodot Fractionation on the Performance of Sensitized Mesoporous Titania Based Photovoltaic Devices. J. Mater. Chem. C 2022, 10, 8824–8833. (link)
Heaney, M.; Adhikari, L.; Siegel, A. L.; Pekar, K.; Lefton, J.; Lamberti, C.; Rungthanapathsophon, P.; Walensky, J. R.; Baker, G. A.; Runčevski, T. Deep Eutectic Solvents Comprising Creatine and Citric Acid and Their Hydrated Mixtures. Chem. Commun. 2022, 58, 2838–2841. (link)
Bhawawet, N.; Larm, N. E.; Adhikari, L.; Polo-Parada, L.; Gutiérrez-Juárez, G.; Baker, G. A. Laser-Induced Sound Pinging for the Rapid Determination of Total Sugar or Sweetener Content in Commercial Beverages. Talanta, 2022, 240, 123034. (link)

Zhao, H.; Martin, C. J.; Larm, N. E.; Baker, G. A.; Trujillo, T. C. Enzyme Activation by Water-Mimicking Dual-Functionalized Ionic Liquids. Mol. Catal. 2021, 515, 111882. (link)
Boogaart, D. J.; Essner, J. B.; Baker, G. A. Evaluation of Canonical Choline Chloride Based Deep Eutectic Solvents as Dye-Sensitized Solar Cell Electrolytes. J. Chem. Phys. 2021, 155, 061102. (link)
Shao, L.; Hu, X.; Sikligar, K.; Baker, G. A.; Atwood, J. L. Coordination Polymers Constructed from Pyrogallol[4]arene-Assembled Metal–Organic Nanocapsules. Acc. Chem. Res. 2021, 54, 3191–3203. (link)
![Coordination Polymers Constructed from Pyrogallol[4]arene-Assembled Metal–Organic Nanocapsules](/sites/default/files/inline-images/li-acc-chem-res.png)
LaRocca, M. M.; Baker, G. A.; Heitz, M. P. Assessing Rotation and Solvation Dynamics in Ethaline Deep Eutectic Solvent and its Solutions with Methanol. J. Chem. Phys. 2021, 155, 034505. (link)
Siegel, A. L.; Baker, G. A. Bespoke Nanostars: Synthetic Strategies, Tactics, and Uses for Tailored Branched Gold Nanoparticles. Nanoscale Advances 2021, 3, 3980–4004. (link)

Larm, N. E.; Adhikari, L.; McKee, S.; Baker, G. A. Polyionic Nanoclays: Tailorable Hybrid Organic-Inorganic Catalytic Platforms. Chem. Mater. 2021, 33, 3585–3592. (link)
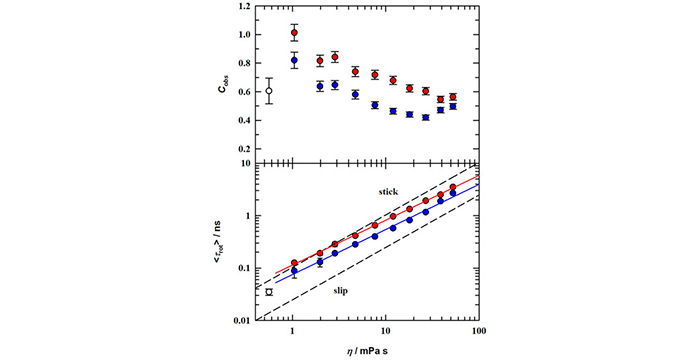
Harris, M. A.; Kinsey, T.; Wagle, D. V.; Baker, G. A.; Sangoro, J. Evidence of a Liquid-Liquid Transition in a Glass-forming Ionic Liquid. Proc. Natl. Acad. Sci. 2021, 118, e2020878118. (link)
Hansen, B. B.; Spittle, S.; Chen, B.; Poe, D.; Zhang, Y.; Klein, J. M.; Horton, A.; Adhikari, L.; Zelovich, T.; Doherty, B. W.; Gurkan, B.; Maginn, E. J.; Ragauskas, A.; Dadmun, M.; Zawodzinski, T. A.; Baker, G. A.; Tuckerman, M. E.; Savinell, R. F.; Sangoro, J. R. Deep Eutectic Solvents: A Review of Fundamentals and Applications. Chem. Rev. 2021, 121, 1232–1285. (link)

Kist, J. A.; Zhao, H.; Mitchell-Koch, K. R.; Baker, G. A. The Study and Application of Biomolecules in Deep Eutectic Solvents. J. Mater. Chem. B 2021, 9, 536–566. (link)

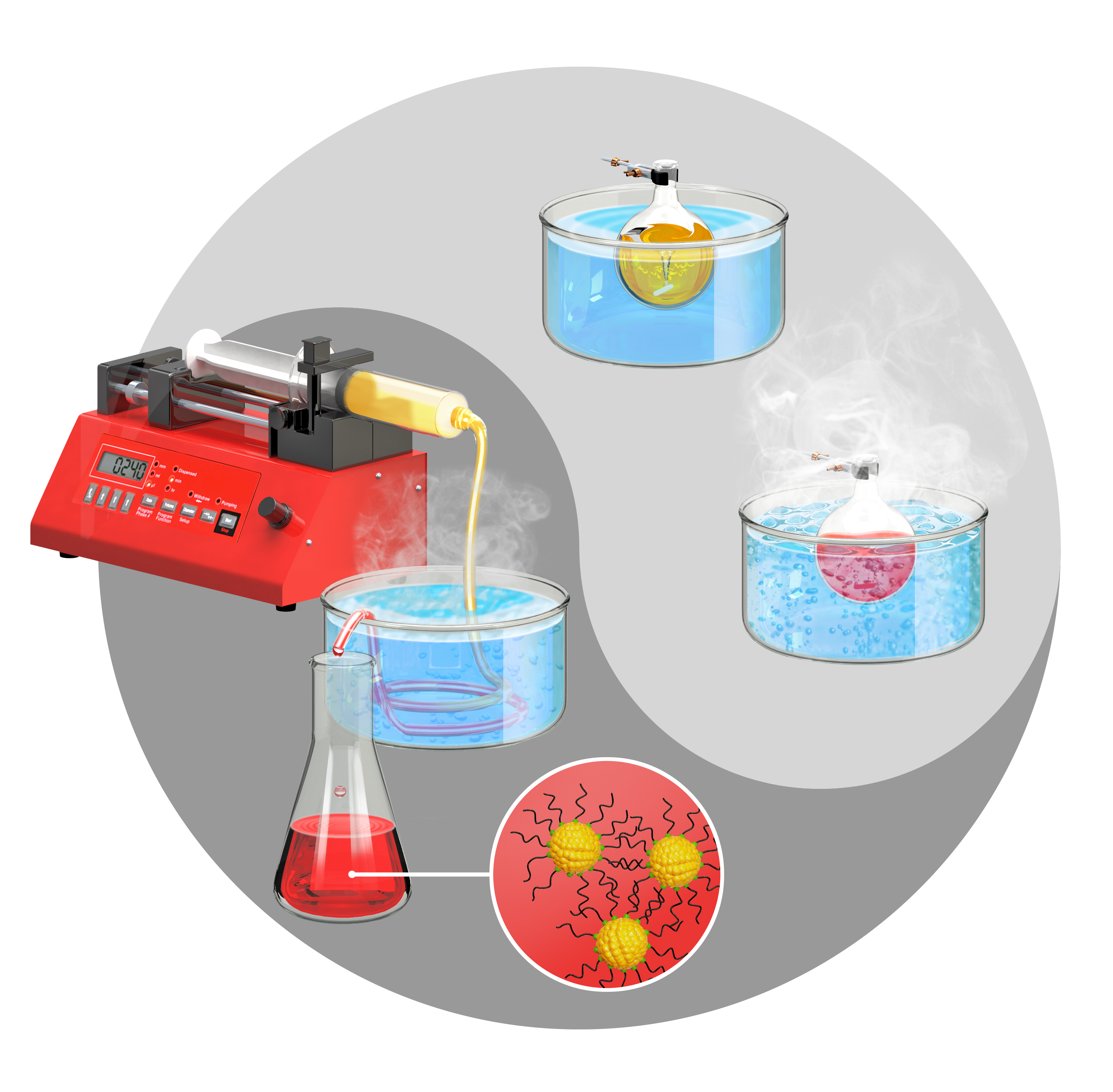
Adhikari, L.; Larm, N. E.; Baker, G. A. Batch and Flow Nanomanufacturing of Large Quantities of Colloidal Silver and Gold Nanocrystals Using Deep Eutectic Solvents. ACS Sustainable Chem. Eng. 2021, 8, 14679–14689. (link)
Larm, N. E.; Essner, J. B.; Thon, J. A.; Bhawawet, N.; Adhikari, L.; St. Angelo, S. K.; Baker, G. A. Single Laboratory Experiment Integrating the Synthesis, Optical Characterization, and Nanocatalytic Assessment of Gold Nanoparticles. J. Chem. Educ. 2020, 97,1454–1459. (link)
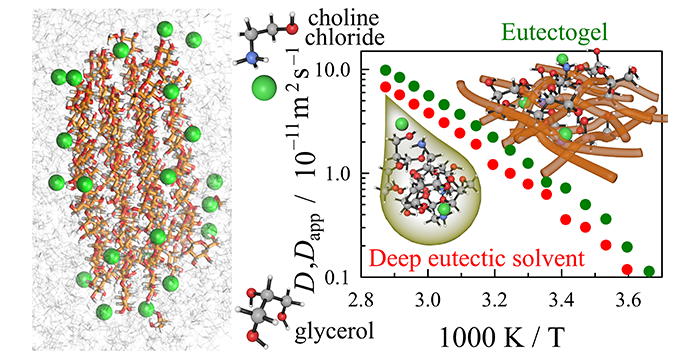
Smith, C. J.; Wagle, D. V.; Bhawawet, N.; Gehrke, S.; Hollóczki, O.; Pingali, S. V.; O’Neill, H.; Baker, G. A. Combined Small-Angle Neutron Scattering, Diffusion NMR, and Molecular Dynamics Study of a Eutectogel: Illuminating the Dynamical Behavior of Glyceline Confined in Bacterial Cellulose Gels. J. Phys. Chem. B 2020, 35, 7647–7658. (link)
Wagle, D. V.; Kelley, S. P.; Sikligar, K.; Baker, G. A.; Atwood, J. L. An Indium‐Seamed Hexameric Metal–Organic Cage as an Example of a Hexameric Pyrogallol[4]arene Capsule Conjoined Exclusively by Trivalent Metal Ions. Angew. Chem. Int. Ed. 2020, 59, 8062–8065. (link)
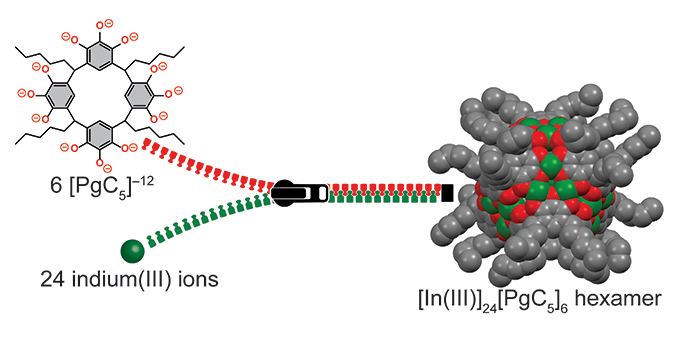
Trivedi, S.; Ravula, S.; Baker, G. A.; Pandey, S.; Bright, F. V. Controlling Microarray Feature Spreading and Response Stability on Porous Silicon Platforms by Using Alkene-Terminal Ionic Liquids and UV Hydrosilylation. Langmuir 2020, 36, 5474–5482. (link)
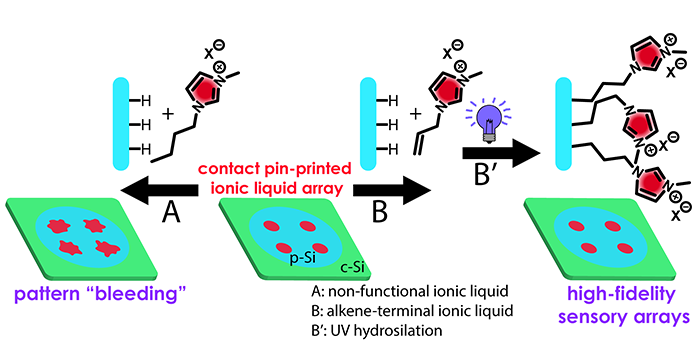
Adhikari, L.; Larm, N. E.; Baker, G. A. Argentous Deep Eutectic Solvent Approach for Scaling Up the Production of Colloidal Silver Nanocrystals. ACS Sustainable Chem. Eng. 2019, 7, 11036–11043. (link)
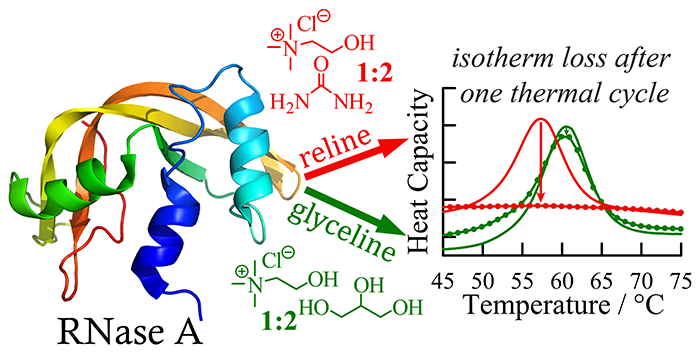
Kist, J. A.; Henzl, M. T.; Bañuelos, J. L; Baker, G. A. Calorimetric Evaluation of the Operational Thermal Stability of Ribonuclease A in Hydrated Deep Eutectic Solvents. ACS Sustainable Chem. Eng. 2019, 7, 12682–12687. (link)
Larm, N. E.; Thon, J. A.; Vazmitsel, Y.; Atwood, J. L.; Baker, G. A. Borohydride stabilized gold–silver bimetallic nanocatalysts for highly efficient 4-nitrophenol reduction. Nanoscale Adv. 2019, 1, 4665–4668. (link)
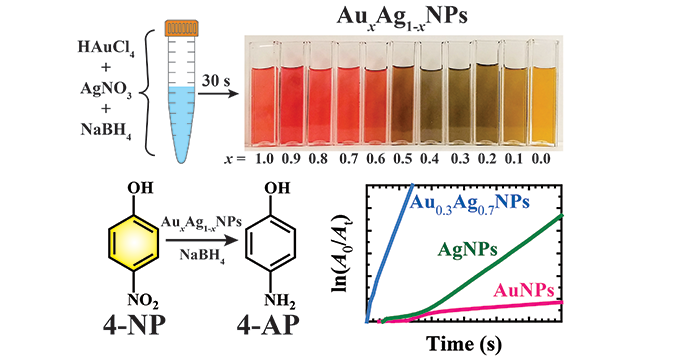
Saladin, M.; Rumble, C. A.; Wagle, D. V.; Baker, G. A.; Maroncelli, M. Characterization of a New Electron Donor–Acceptor Dyad in Conventional Solvents and Ionic Liquids. J. Phys. Chem. B 2019, 123, 44, 9395–9407. (link)
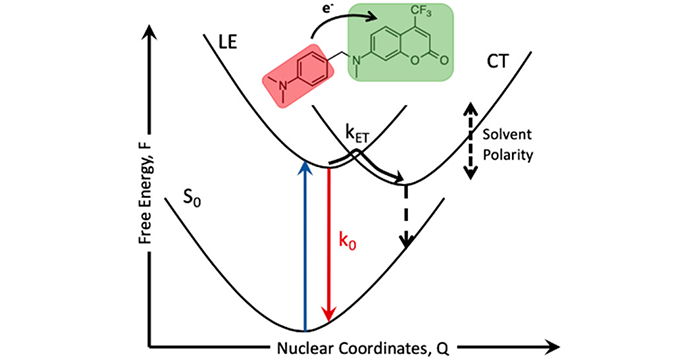
Larm, N. E.; Bhawawet, N.; Thon, J. A.; Baker, G. A. Best practices for reporting nanocatalytic performance: lessons learned from nitroarene reduction as a model reaction. New J. Chem. 2019, 43, 17932–17936. (link)
Larm, N. E.; Madugula, D.; Lee, M. W.; Baker, G. A. Polyhedral borane-capped coinage metal nanoparticles as high-performing catalysts for 4-nitrophenol reduction. Chem. Commun. 2019, 55, 7990–7993. (link)
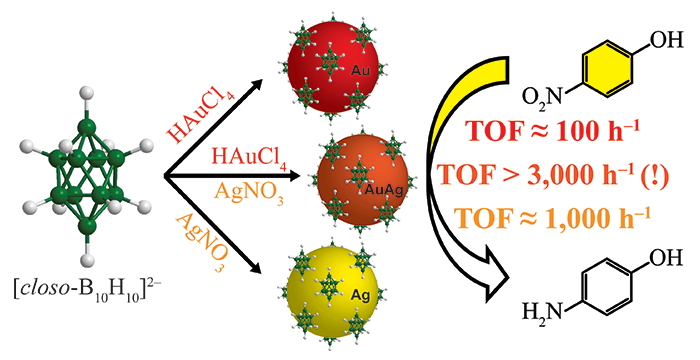
Thon, J. A.; Larm, N. E.; Vazmitsel, Y.; Baker, G. A. Plasmonic Evolution and Arrested Development for Silver Nanoscale Colloids: A Classroom Demonstration. J. Chem. Educ. 2019, 96, 11, 2560–2564. (link)
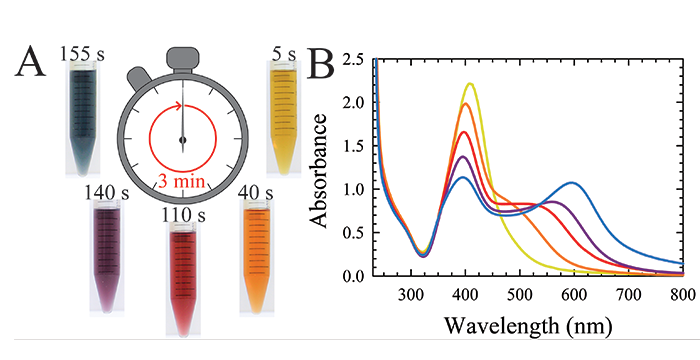
Ravula, S.; Larm, N. E.; Mottaleb, M. A.; Heitz, M. P.; Baker, G. A. Vapor Pressure Mapping of Ionic Liquids and Low-Volatility Fluids Using Graded Isothermal Thermogravimetric Analysis. ChemEngineering 2019, 3, 42 (12pp). (link)
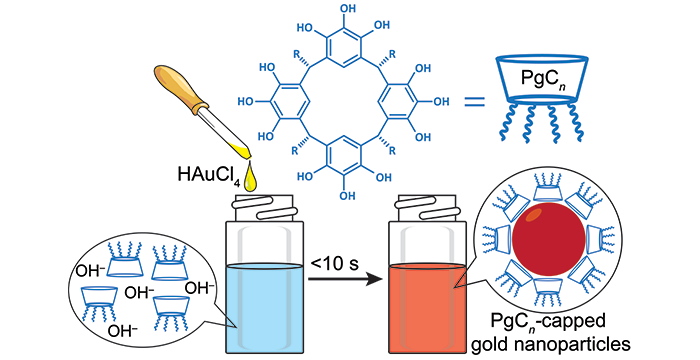
Adhikari, L.; Larm, N. E.; Wagle, D. V.; Atwood, J. L.; Baker, G. A. Facile, one-pot, in aqua synthesis of catalytically competent gold nanoparticles using pyrogallol[4]arene as the sole reagent. Chem. Commun. 2019, 55, 6261–6264. (link)
Essner, J. B.; Kist, J. A.; Polo-Parada, L.; Baker, G. A. Artifacts and Errors Associated with the Ubiquitous Presence of Fluorescent Impurities in Carbon Nanodots. Chem. Mater. 2018, 30, 1878–1887. (link)

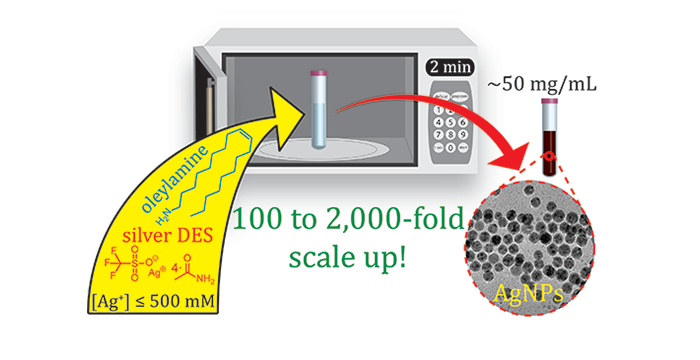
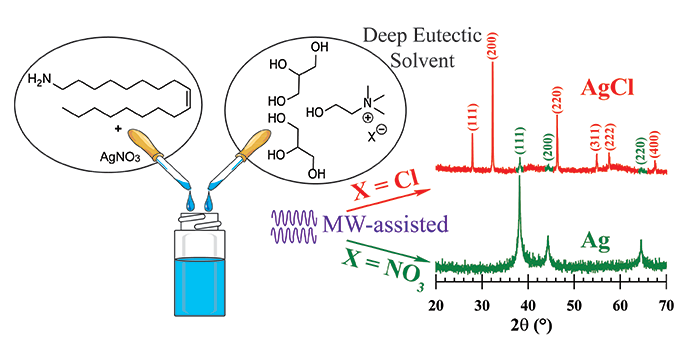
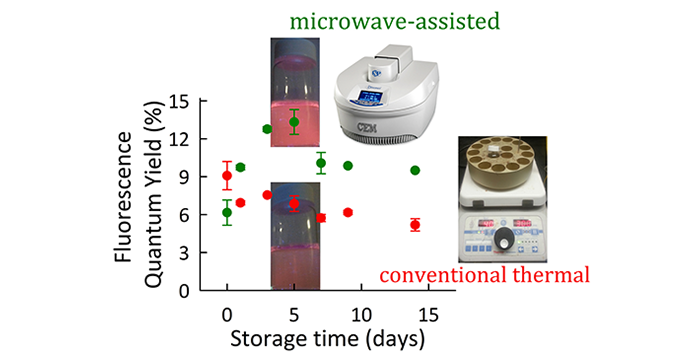
Adhikari, L.; Larm, N. E.; Bhawawet, N.; Baker, G. A. Rapid Microwave-Assisted Synthesis of Silver Nanoparticles in a Halide-Free Deep Eutectic Solvent. ACS Sustainable Chem. Eng, 2018, 6, 5725–5731. (link)
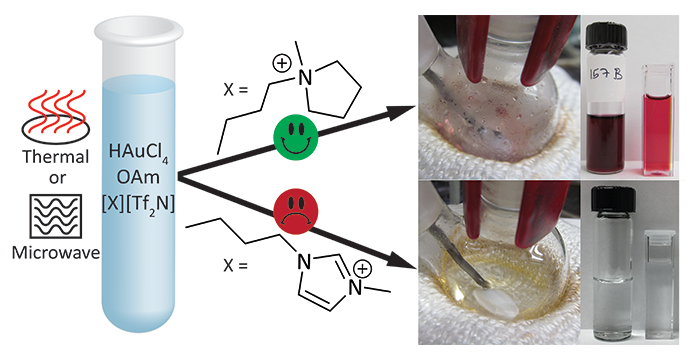
Bhawawet, N.; Essner, J. B.; Atwood, J. L.; Baker, G. A. On the non-innocence of the imidazolium cation in a rapid microwave synthesis of oleylamine-capped gold nanoparticles in an ionic liquid. Chem. Commun. 2018, 54, 7523–7526. (link)
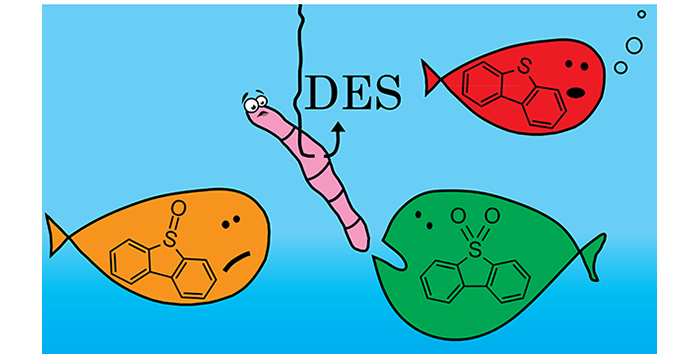
Wagle, D. V.; Zhao, H.; Deakyne, C. A.; Baker, G. A. Quantum Chemical Evaluation of Deep Eutectic Solvents for the Extractive Desulfurization of Fuel. ACS Sustainable Chem. Eng. 2018, 6, 7525–7531. (link)
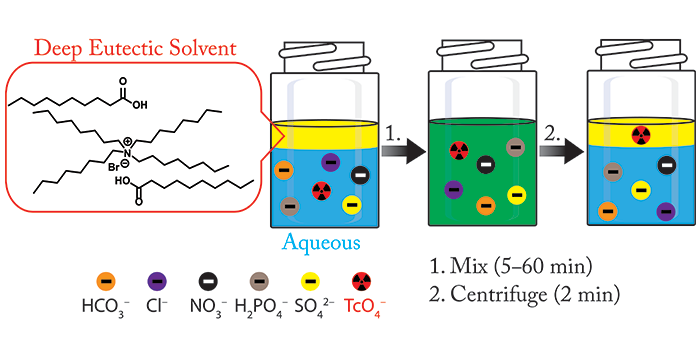
Phelps, T. E.; Bhawawet, N.; Jurisson, S. S.; Baker, G. A. Efficient and Selective Extraction of 99mTcO4– from Aqueous Media using Hydrophobic Deep Eutectic Solvents. ACS Sustainable Chem. Eng. 2018, 6, 13656–13661. (link)
Zhang, C.; Sikligar, K.; Patil, R. S.; Barnes, C. L.; Baker, G. A.; Atwood, J. L. A M18L6 metal–organic nanocapsule with open windows using mixed macrocycles. Chem. Commun. 2018, 54, 635–637. (link)
Shen, X.; Laber, C. H.; Sarkar, U.; Galazzi, F.; Johnson, K. M.; Mahieu, N. G.; Hillebrand, R.; Fuchs-Knotts, T.; Barnes, C. L.; Baker, G. A.; Gates, K. S. Exploiting the Inherent Photophysical Properties of the Major Tirapazamine Metabolite in the Development of Profluorescent Substrates for Enzymes That Catalyze the Bioreductive Activation of Hypoxia-Selective Anticancer Prodrugs. J. Org. Chem. 2018, 83, 3126–3131. (link)

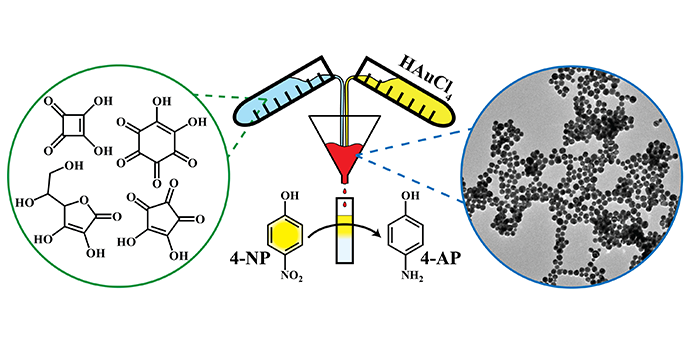
Larm, N. E.; Essner, J. B.; Pokpas, K.; Canon, J. A.; Jahed, N.; Iwuoha, E. I.; Baker, G. A. Room-Temperature Turkevich Method: Formation of Gold Nanoparticles at the Speed of Mixing Using Cyclic Oxocarbon Reducing Agents. J. Phys. Chem. C 2018, 122, 5105–5118. (link)
Smith, C. J.; Wagle, D. V.; O'Neill, H. M.; Evans, B. R.; Baker, S. N.; Baker, G. A. Bacterial Cellulose Ionogels as Chemosensory Supports. ACS Appl. Mater. Interfaces 2017, 9, 43, 38042–38051. (link)

Wagle, D.V.; Rondinone, A. J.; Woodward, J. D.; Baker, G. A. Polyol Synthesis of Magnetite Nanocrystals in a Thermostable Ionic Liquid. Cryst. Growth Des. 2017, 17, 1558–1567. (link)

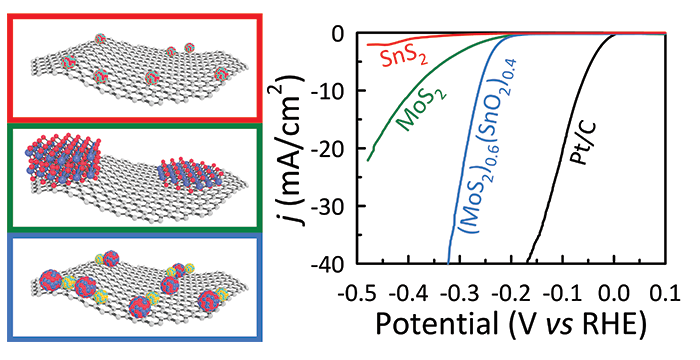
Ravula, S.; Zhang, C.; Essner, J. B.; Robertson, J. D.; Lin, J.; Baker, G. A. Ionic Liquid-Assisted Synthesis of Nanoscale (MoS2)x(SnO2)1–x on Reduced Graphene Oxide for the Electrocatalytic Hydrogen Evolution Reaction. ACS Appl. Mater. Interfaces 2017, 9, 8065–8074. (link)
Essner, J. B.; Baker, G. A. The emerging roles of carbon dots in solar photovoltaics: a critical review. Environ. Sci.: Nano 2017, 4, 1216–1263. (link)
Rumble, C. A.; Kaintz, A.; Yadav, S. K.; Conway, B.; Araque, J. C.; Baker, G. A.; Margulis, C.; Maroncelli, M. Rotational Dynamics in Ionic Liquids from NMR Relaxation Experiments and Simulations: Benzene and 1-Ethyl-3-Methylimidazolium. Phys. Chem. B 2016, 120, 9450–9467. (link)
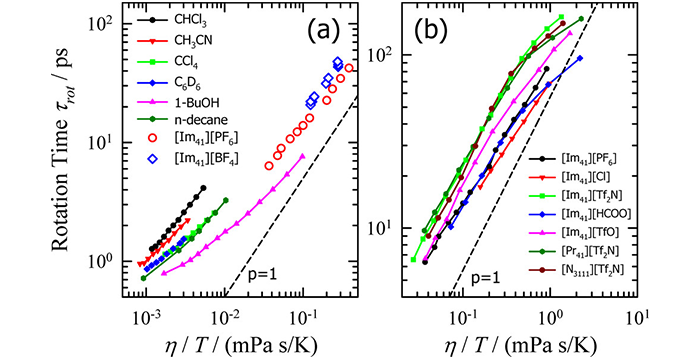
Zhao, H.; Baker, G. A.; Wagle, D. V.; Ravula, S.; Zhang, Q. Tuning Task-Specific Ionic Liquids for the Extractive Desulfurization of Liquid Fuel. ACS Sustainable Chem. Eng. 2016, 4, 4771–4780. (link)
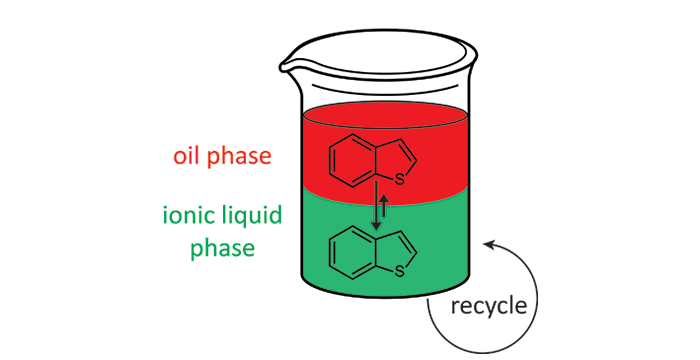
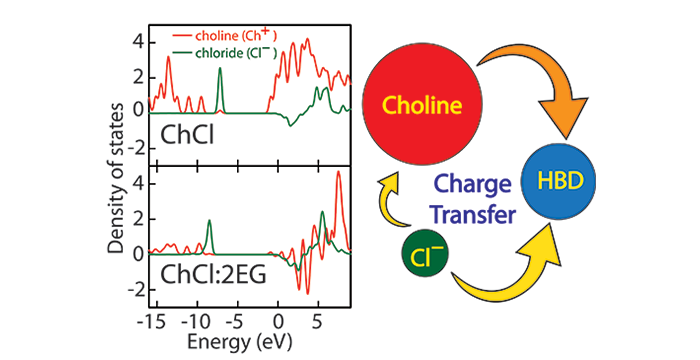
Wagle, D. V.; Deakyne, C. A.; Baker, G. A. Quantum Chemical Insight into the Interactions and Thermodynamics Present in Choline Chloride Based Deep Eutectic Solvents. J. Phys. Chem. B 2016, 120, 6739–6746. (link)
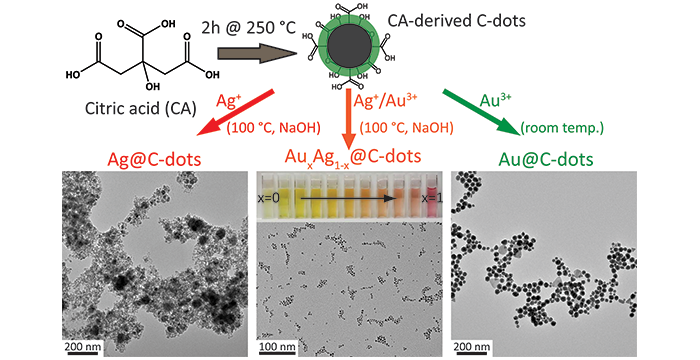
Essner, J. B.; Laber, C. H.; Baker, G. A. Effective size and optical tailoring of catalytic bimetallic nanoparticles generated from citric acid-derived carbon nanodots. J. Mater. Chem A 2015, 3, 16354–16360. (link)
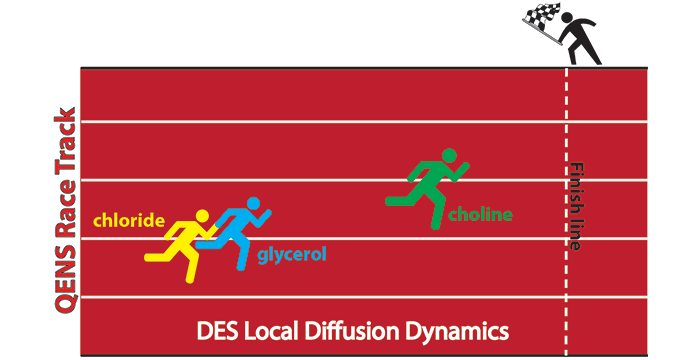
Wagle, D. V.; Baker, G. A.; Mamontov, E. Differential Microscopic Mobility of Components within a Deep Eutectic Solvent. J. Phys. Chem. Lett. 2015, 6, 2924–2928. (link)
Ravula, S.; Essner, J. B.; Baker, G. A. Kitchen-Inspired Nanochemistry: Dispersion, Exfoliation, and Hybridization of Functional MoS2 Nanosheets using Culinary Hydrocolloids. ChemNanoMat 2015, 1, 167–177. (link)

Ravula, S.; Baker, S. N.; Kamath, G.; Baker, G. A. Ionic liquid-assisted exfoliation and dispersion: stripping graphene and its two-dimensional layered inorganic counterparts of their inhibitions. Nanoscale 2015, 7, 4338–4353. (link)
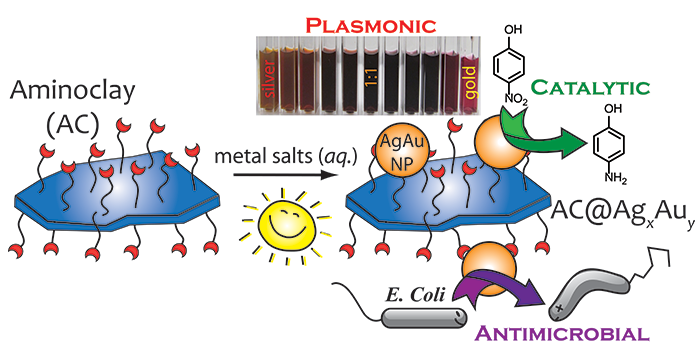
Ravula, S.; Essner, J. B.; La, W. A.; Polo-Parada, L.; Kargupta, R.; Hull, G. J.; Sengupta, S.; Baker, G. A. Sunlight-assisted route to antimicrobial plasmonic aminoclay catalysts. Nanoscale 2015, 7, 86–91. (link)
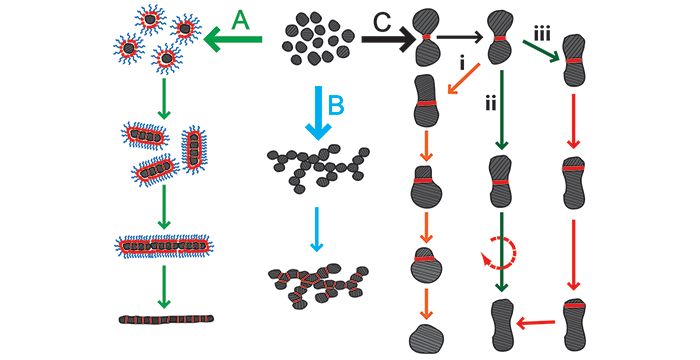
Wagle, D. V.; Baker, G. A. Cold welding: a phenomenon for spontaneous self healing and shape genesis at the nanoscale. Mater. Horiz. 2015, 2, 157–167. (link)
Wagle, D. V.; Zhao, H.; Baker, G. A. Deep Eutectic Solvents: Sustainable Media for Nanoscale and Functional Materials. Acc. Chem. Res. 2014, 47, 2299–2308. (link)
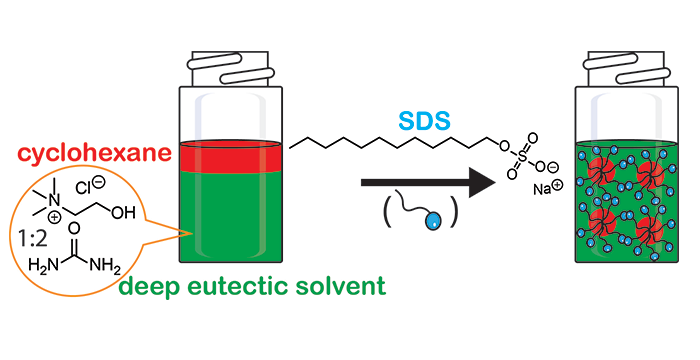
Pal, M.; Rai, R.; Yadav, A.; Khanna, R.; Baker, G. A.; Pandey, S. Self-Aggregation of Sodium Dodecyl Sulfate within (Choline Chloride + Urea) Deep Eutectic Solvent. Langmuir 2014, 30, 13191–13198. (link)
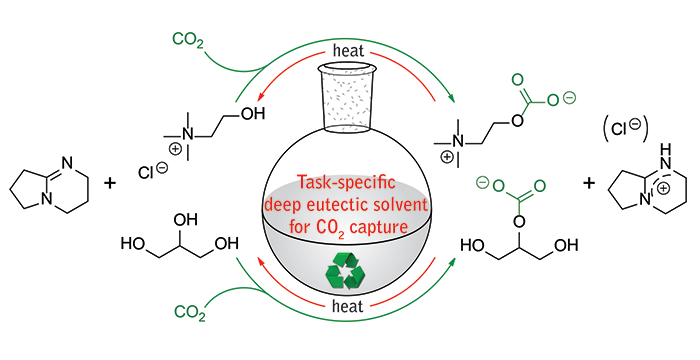
Sze, L. L.; Pandey, S.; Ravula, S.; Pandey, S.; Zhao, H.; Baker, G. A.; Baker, S. N. Ternary Deep Eutectic Solvents Tasked for Carbon Dioxide Capture. ACS Sustainable Chem. Eng. 2014, 2, 2117–2123. (link)
Chen, X.; Essner, J. B.; Baker, G. A. Exploring luminescence-based temperature sensing using protein-passivated gold nanoclusters. Nanoscale 2014, 6, 9594–9598. (link)

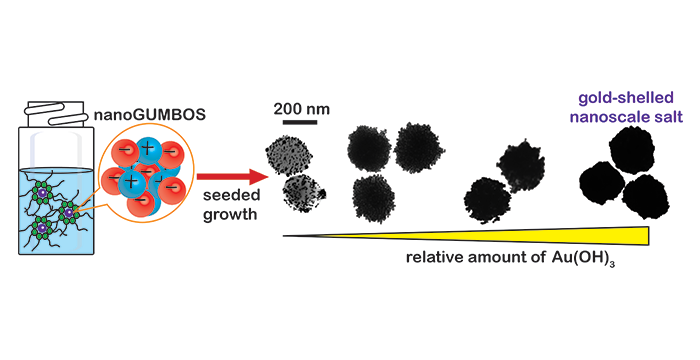
Wright, A. R.; Li, M.; Ravula, S.; Cadigan, M.; El-Zahab, B.; Das, S.; Baker, G. A.; Warner, I. M. Soft- and Hard-Templated Organic Salt Nanoparticles with the Midas Touch: Gold-Shelled nanoGUMBOS. J. Mater. Chem. C 2014, 2, 8996–9003. (link)
Ramirez-Perez, F. I.; Gutiérrez-Juárez, G.; Bok, S.; Gangopadhyay, K.; Gangopadhyay, S.; Baker, G. A.; Polo-Parada, L. Dye-Doped Organosilicate Nanoparticles as Cell-Preserving Labels for Photoacoustic Signal Generation. J. Biomed. Nanotechnol. 2014, 10, 3337–3350. (link)
Hofmann, C. M.; Essner, J. B.; Baker, G. A.; Baker, S. N. Protein-templated gold nanoclusters sequestered within sol–gel thin films for the selective and ratiometric luminescence recognition of Hg2+. Nanoscale 2014, 6, 5425–5431. (link)
Xia, S.; Baker, G. A.; Li, H.; Ravula, S.; Zhao, H. Aqueous ionic liquids and deep eutectic solvents for cellulosic biomass pretreatment and saccharification. RSC Adv. 2014, 4, 10586–10596. (link)
Zhao, H.; Baker, G. A. Ionic Liquids and Deep Eutectic Solvents for Biodiesel Synthesis: a Review. J. Chem. Technol. Biotechnol. 2013, 88, 3–12. (link)
Fowler, D. A.; Atwood, J. L.; Baker, G. A. Formation of a dimeric host–guest complex via binding between a dicationic ionic liquid and a pyrogallol[4]arene macrocycle. Chem. Commun. 2013, 49, 1802–1804. (link)
![Formation of a dimeric host–guest complex via binding between a dicationic ionic liquid and a pyrogallol[4]arene macrocycle](/sites/default/files/inline-images/toc-drew-dicat.png)
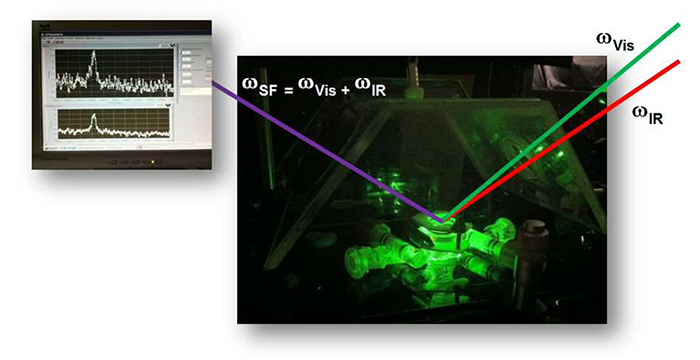
Peñalber, C. Y.; Baker, G. A.; Baldelli, S. Sum Frequency Generation Spectroscopy of Imidazolium-Based Ionic Liquids with Cyano-Functionalized Anions at the Solid Salt–Liquid Interface. J. Phys. Chem. B 2013, 117, 5939–5949. (link)
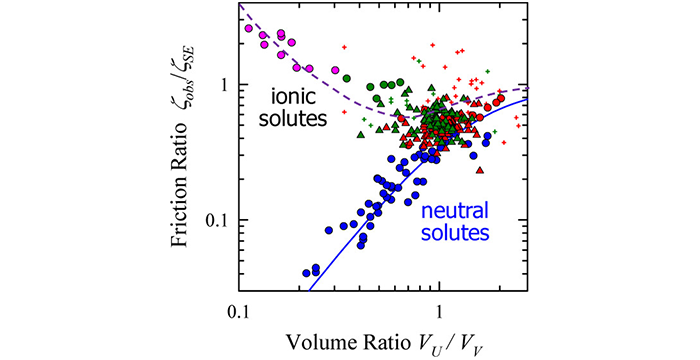
Kaintz, A.; Baker, G.; Benesi, A.; Maroncelli, M. Solute Diffusion in Ionic Liquids, NMR Measurements and Comparisons to Conventional Solvents. J. Phys. Chem. B 2013, 117, 11697–11708. (link)
Chen, X.; Baker, G. A. Cholesterol determination using protein-templated fluorescent gold nanocluster probes. Analyst 2013, 138, 7299–7302. (link)
Tang, S.; Baker, G. A.; Zhao, H. Ether- and alcohol-functionalized task-specific ionic liquids: attractive properties and applications. Chem. Soc. Rev. 2012, 41, 4030–4066. (link)
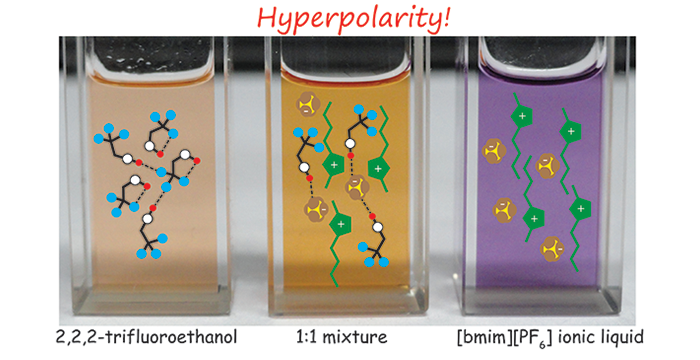
Trivedi, S.; Pandey, S.; Baker, S. N.; Baker, G. A. Pronounced Hydrogen Bonding Giving Rise to Apparent Probe Hyperpolarity in Ionic Liquid Mixtures with 2,2,2-Trifluoroethanol. J. Phys. Chem. B 2012, 116, 1360–1369. (link)
Tang, S.; Baker, G. A.; Ravula, S.; Jones, J. E.; Zhao, H. PEG-functionalized ionic liquids for cellulose dissolution and saccharification. Green Chem. 2012, 14, 2922–2932. (link)
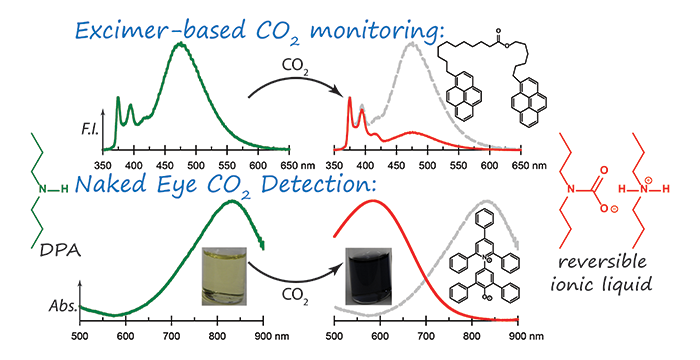
Pandey, S.; Baker, S. N.; Pandey, S.; Baker, G. A. Optically responsive switchable ionic liquid for internally-referenced fluorescence monitoring and visual determination of carbon dioxide. Chem. Commun. 2012, 48, 7043–7045. (link)
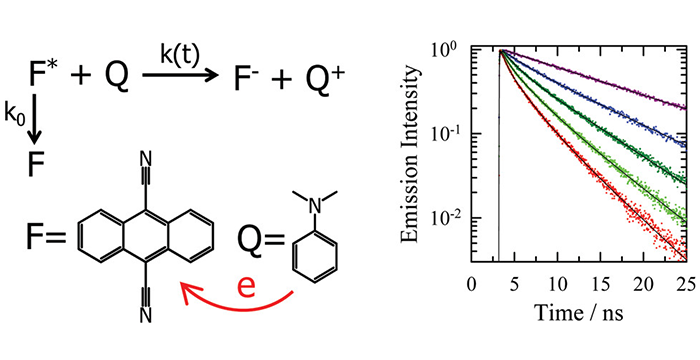
Liang, M.; Kaintz, A.; Baker, G. A.; Maroncelli, M. Bimolecular Electron Transfer in Ionic Liquids: Are Reaction Rates Anomalously High? J. Phys. Chem. B 2012, 116, 1370–1384. (link)
Jordan, A. N.; Das, S.; Siraj, N.; De Rooy, S. L.; Li, M.; El-Zahab, B.; Chandler, L.; Baker, G. A.; Warner, I. M. Anion-controlled morphologies and spectral features of cyanine-based nanoGUMBOS — an improved photosensitizer. Nanoscale 2012, 4, 5031–5038. (link)
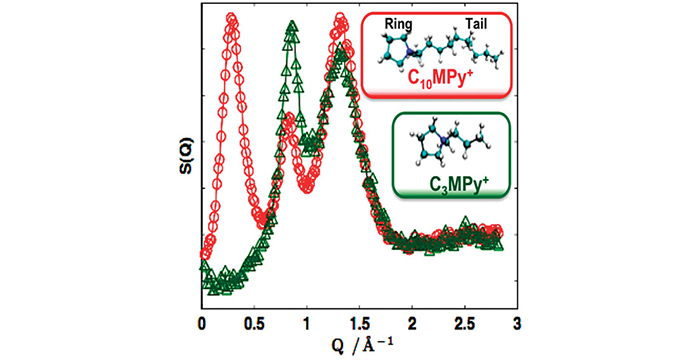
Li, S.; Bañuelos, J. L.; Guo, J.; Anovitz, L.; Rother, G.; Shaw, R. W.; Hillesheim, P. C.; Dai, S.; Baker, G. A.; Cummings, P. T. Alkyl Chain Length and Temperature Effects on Structural Properties of Pyrrolidinium-Based Ionic Liquids: A Combined Atomistic Simulation and Small-Angle X-Ray Scattering Study. J. Phys. Chem. Lett. 2012, 3, 125–130. (link)
Al-Azzawi, O. M.; Hofmann, C. M.; Baker, G. A.; Baker, S. N. Nanosilica-supported polyethoxyamines as low-cost, reversible carbon dioxide sorbents. J. Colloid Interface Sci. 2012, 385, 154–159. (link)
Bok, S.; Korampally, V.; Polo-Parada, L.; Mamidi, V.; Baker, G. A.; Gangopadhyay, K.; Folk, W. R.; Dasgupta, P. K.; Gangopadhyay, S. Confeito-Like Assembly of Organosilicate-Caged Fluorophores: Ultrabright Suprananoparticles for Fluorescence Imaging. Nanotechnology 2012, 23, 175601 (11pp). (link)
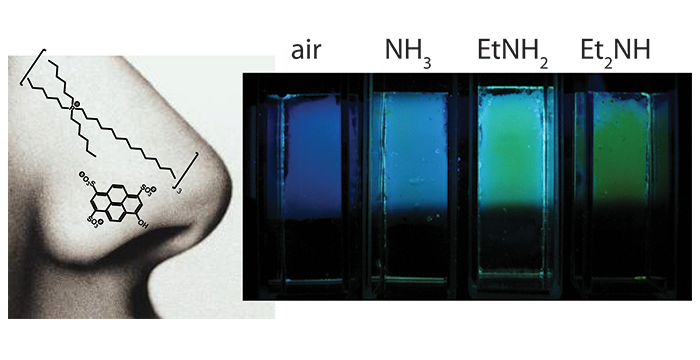
Yung, K. Y.; Schadock-Hewitt, A. J.; Hunter, N. P.; Bright, F. V.; Baker, G. A. 'Liquid litmus': chemosensory pH-responsive photonic ionic liquids. Chem. Commun. 2011, 47, 4775–4777. (link)