
Research in the Bernskoetter lab focuses on the use of inorganic and organometallic complexes to address challenges relevant to our planet's growing energy concerns. Our initiatives employ techniques from synthetic organic and inorganic chemistry to study highly reactive molecules capable of mediating difficult chemical transformations.
Education:
Prof. Bernskoetter received the B.Sc. in Chemisty from Benedictine College, Atchison, KS in 2002 and the Ph.D. in Chemistry from Cornell University in 2006.
Professional Experience:
Prof. Bernskoetter began his postgraduate research work as a Postdoctoral Research Fellow at the University of North Carolina-Chapel Hill where he worked with Prof. Maurice Brookhart where he studied carbon-hydrogen bond activation. In 2009 he began his academic career at Brown University where he became the Manning Assistant Professor of Chemistry in 2012. In 2015, he returned to his home state and the University of Missouri.
Honors and Awards:
2014 Alfred P. Sloan Research Fellow in Chemistry; Alfred P. Sloan Foundation, New York, NY
2014 National Science Foundation Early Career Award; NSF, Arlington, VA
2012 Awarded Manning Assistant Professor of Chemistry chair; Brown University, Providence, RI
2010 Air Force Office of Scientific Research Young Investigator Award; AFOSR, Arlington, VA
2009 Richard B. Salomon Faculty Research Award; Brown University, Providence, RI
2006 Tunis Wentink Thesis of the Year Award; Cornell University, Ithaca, NY
Research:
(1) Carbon Dioxide Functionalization for Fuels. As world-wide petrochemical reserves increase in scarcity, chemists must endeavor to find renewable and economical alternatives to fossil fuel carbon sources. Many potential clean energy sources, such as hydrogen, have significant problems with storage and transport. Our laboratory seeks new catalysts to fix hydrogen and carbon dioxide into safe and convenient liquid fuels. In route to this goal, we hope to build our fundumental understanding of the feature that control transition metal reactions.
(2) Renewable Feedstocks for Commodity Chemicals. Much as our fuel economy depends on fossil fuels, our chemical economy relies heavily on non-renewable petroleum feedstocks. Carbon dioxide offers are cheap, non-toxic, renewable, but virtually untapped resource for creating more sustainable commodity chemicals. Our program explores new transition metal catalysts which can create value-added chemicals with existing markets for carbon dioxide and other sustainable resources.

Townsend, Tanya M.; Bernskoetter, Wesley H.; Hazari, Nilay; Mercado, Brandon Q. “Dehydrogenative Synthesis of Carbamates from Formamides and Alcohols Using a Pincer-Supported Iron Catalyst” ACS Catalysis (2021), 11(16), 10614-10624. 10.1021/acscatal.1c02718.
Adamson, Tristan T.; Kelley, Steven P.; Bernskoetter, Wesley H. “Iron-Mediated C-C Bond Formation via Reductive Coupling with Carbon Dioxide” Organometallics (2020), 39(19), 3562-3571. 10.1021/acs.organomet.0c00528
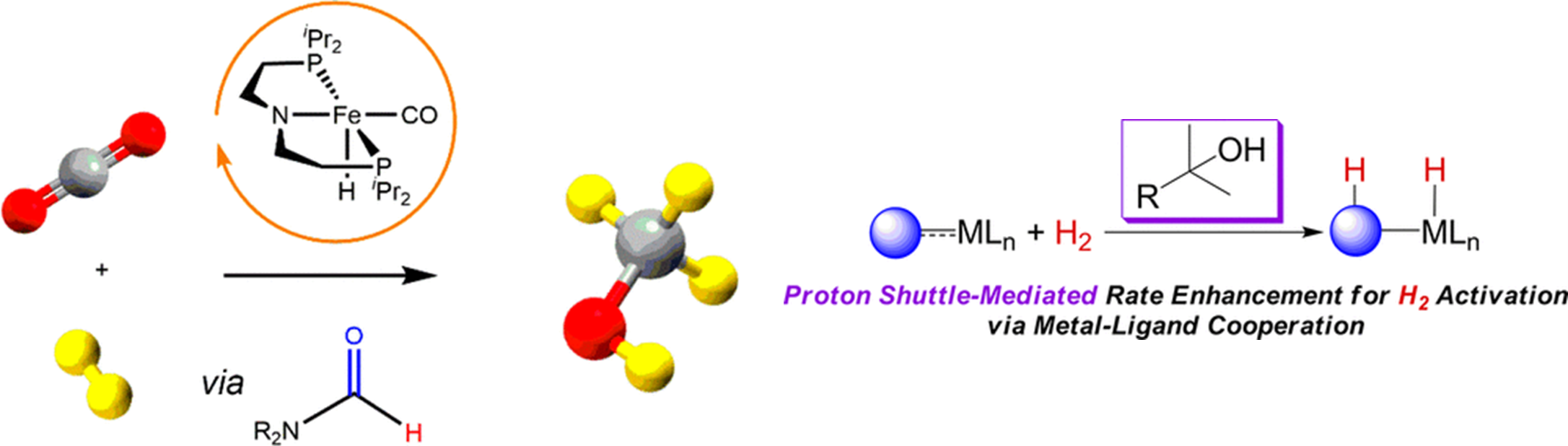
Smith, Nicholas E.; Bernskoetter, Wesley H.; Hazari, Nilay. “The Role of Proton Shuttles in the Reversible Activation of Hydrogen via Metal-Ligand Cooperation” Journal of the American Chemical Society (2019), 141(43), 17350-17360. 10.1021/jacs.9b09062
Lane, Elizabeth M.; Zhang, Yuanyuan; Hazari, Nilay; Bernskoetter, Wesley H. “Sequential Hydrogenation of CO2 to Methanol Using a Pincer Iron Catalyst” Organometallics (2019), 38(15), 3084-3091. 10.1021/acs.organomet.9b00413