
Education:
B.A. Chemistry, New College of Florida, 2005
Ph.D. Chemistry, University of California, Irvine, 2009
Professional Experience:
Department Chair, 2021-
Professor, Mizzou, 2021-
Associate Professor, Mizzou, 2016-2021
Associate Chair for Undergraduate Studies, 2015-2021
Assistant Professor, Mizzou, 2011-2016
Postdoctoral Research Associate, Texas A&M University, 2010
Honors and Awards:
2022-24, Alexander von Humboldt Research Fellowship for Experienced Researchers
2020, Blue Chalk Advising Award, MU College of Arts & Science
2018 & 2019, Fuldner Faculty Fellow, MU Department of Chemistry
2017, MU President's Award for Early Career Excellence
2017, Faculty Fellowship Summer Institute in Israel
2015-2020, Department of Energy Early Career award
2013, Southeastern Conference (SEC) travel grant award
2011-2016, Nuclear Forensics Junior Faculty Award, Department of Homeland Security, Domestic Nuclear Detection Office
2009, Glenn T. Seaborg Institute Fellowship, Los Alamos National Laboratory
2006, Chemistry Graduate Student Teaching Award: Contributions to the Chemistry Department Teaching Program by a First Year Teaching Assistant
Synthesis; Actinide chemistry; Chalcogen and pnictogen chemistry; Density Functional Theory; Group 11 chemistry
Our research is divided into two areas: 1) Structure, bonding, spectroscopy, and reactivity of actinide complexes, and 2) Group 11/Main group chemistry
We are interested in exploring the fundamental nature of actinide-ligand bonding with soft donor ligands such as the heavier chalcogens and pnictogens. Soft donor ligands are relevant to separation methods as sulfur-based ligands have been shown to have more selectivity towards actinides over lanthanides. In our overall goal to provide insight into actinide-ligand bonding we have two objectives: 1) To investigate the molecular and electronic structure of organometallic complexes of thorium, uranium, and neptunium with soft donor ligands such as S, Se, Te, P, and As having sigma (single) bonds as well as sigma and pi (double) bonds. Reactivity patterns are established and compared to their first row counterparts (O and N). 2) The fundamental coordination chemistry and properties of neptunium, especially Np(III) and Np(IV) complexes.
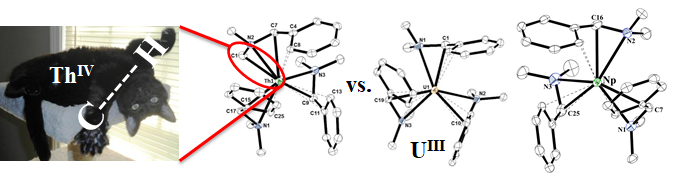
Publications in this area:
Dalton Trans. 2015, 44, 2693 (special issue on f-element chemistry)
J. Am. Chem. Soc. 2015, 137, 14846
Inorg. Chem. 2015, 54, 11625
Dalton Trans. 2016, 45, 10042 (special issue on New Talent: Americas)
Inorg. Chem. 2016, 55, 12675
Chem. Commun. 2016, 52, 14373
Angew. Chem. Int. Ed. 2017, 56, 12925
Chem. Eur. J. 2017, 23, 16748
J. Organomet. Chem. 2018, 857, 159 (special issue for Bill Evans)
Inorg. Chem. 2018, 57, 7270
Organometallics 2018, 37, 1884
Dalton Trans. 2018, 47, 8189
Inorg. Chem. 2018, 57, 10518
Chem. Commun. 2018, 54, 10435
Angew. Chem. Int. Ed. 2018, 57, 16748
Organometallics 2019, 38, 1733
Angew. Chem. Int. Ed. 2019, 58, 14891
Organometallics 2020, 39, 2152
Chem. Sci. 2020, 11, 5830
Inorg. Chem. 2020, 59, 16137
Organometallics 2021, 40, 1411
Inorg. Chem. 2021, 60, 10614
Organometallics 2021, 40, 2701
2) Transition Metal and Main Group Chemistry
Group 11 elements have importance as fluorescent probes, sensors, and biological systems. The reactivity of dinuclear group 11 complexes, especially with amidinate ligands, is being investigated to examine their luminescence, metallophilic interactions, and redox chemistry.
The chemistry of chalcogen and pnictogen elements is also of interest, especially the design of new ligands to support our actinide chemistry or the synthesis of fluorescent compounds in collaboration with Gary Baker's group.
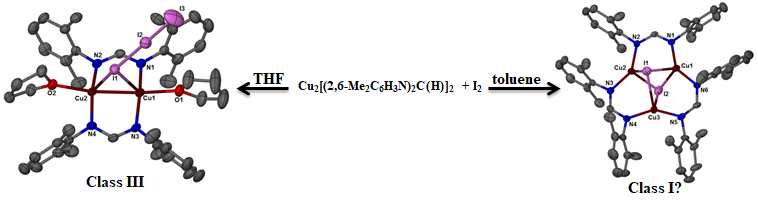
Publications in this area:
Inorganics 2014, 2, 540 (special issue on Frontier in Gold Chemistry)
Inorg. Chem. 2014, 53, 11357
Organometallics 2015, 34, 3 (selected as issue cover)
Inorg. Chem. 2015, 54, 8509
Dalton Trans. 2016, 45, 14265
Dalton Trans. 2017, 46, 10867